Formula of density6/20/2023 In the case of dry sand, sand is so much denser than air that the buoyancy effect is commonly neglected (less than one part in one thousand). If the material is under pressure (commonly ambient air pressure at the earth's surface) the determination of mass from a measured sample weight might need to account for buoyancy effects due to the density of the void constituent, depending on how the measurement was conducted. In the case of non-compact materials, one must also take care in determining the mass of the material sample. In the case of sand, it could be water, which can be advantageous for measurement as the void fraction for sand saturated in water-once any air bubbles are thoroughly driven out-is potentially more consistent than dry sand measured with an air void. In practice, the void fraction is not necessarily air, or even gaseous. It might be loose or compact, with more or less air space depending on handling. Some bulk materials, however, such as sand, have a variable void fraction which depends on how the material is agitated or poured. For the close-packing of equal spheres the non-void fraction can be at most about 74%. Sometimes this can be determined by geometrical reasoning. 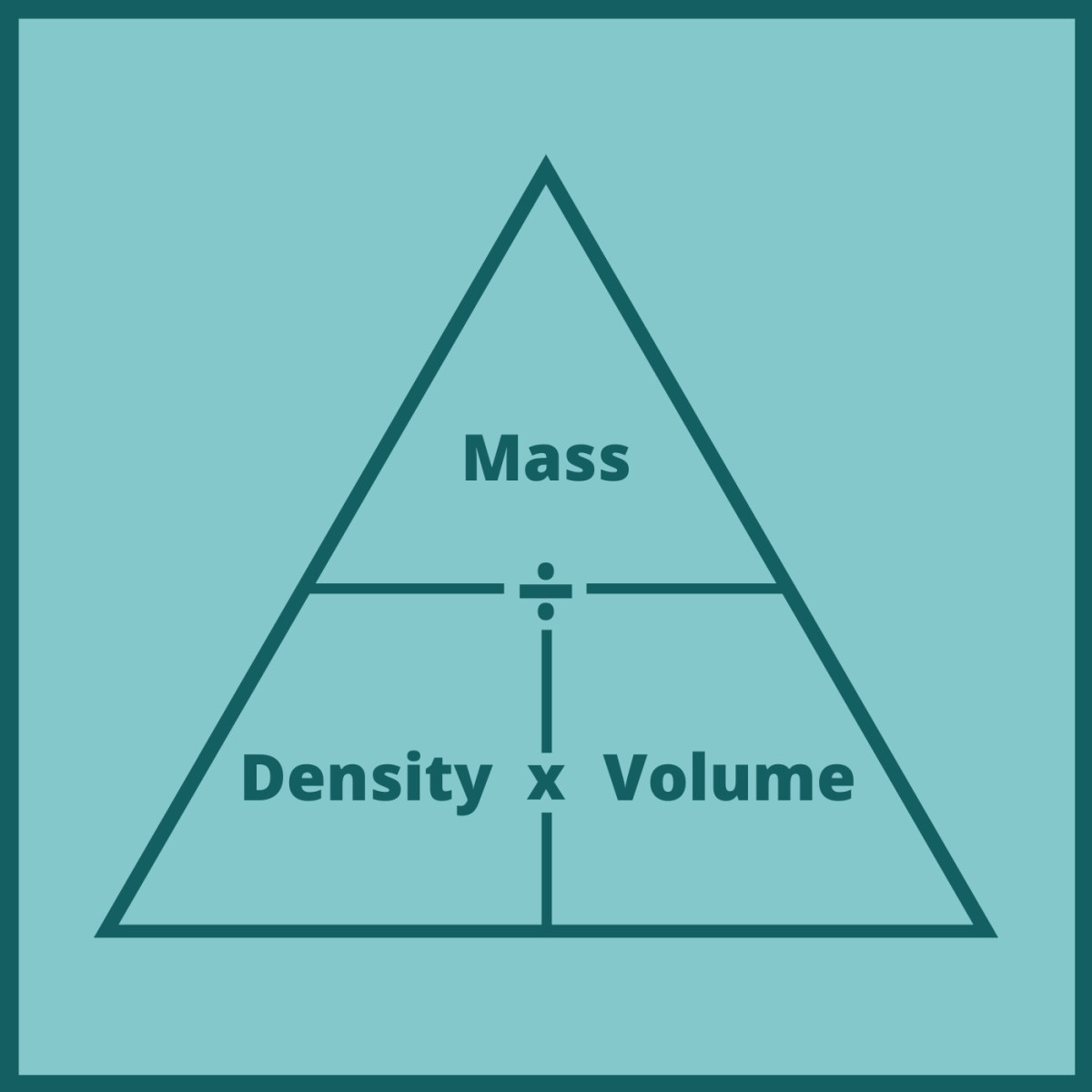
To determine volumetric mass density, one must first discount the volume of the void fraction. This is not the same thing as volumetric mass density. Mass divided by bulk volume determines bulk density. with a calibrated measuring cup) or geometrically from known dimensions. The bulk volume of a material-inclusive of the void fraction-is often obtained by a simple measurement (e.g. Commonly the void is air, but it could also be vacuum, liquid, solid, or a different gas or gaseous mixture. Voids are regions which contain something other than the considered material. Many materials exist in nature as flakes, pellets, or granules. In practice, bulk materials such as sugar, sand, or snow contain voids. The equations continues for metals 3, 4 etc.Ρ = m V Non-compact materials 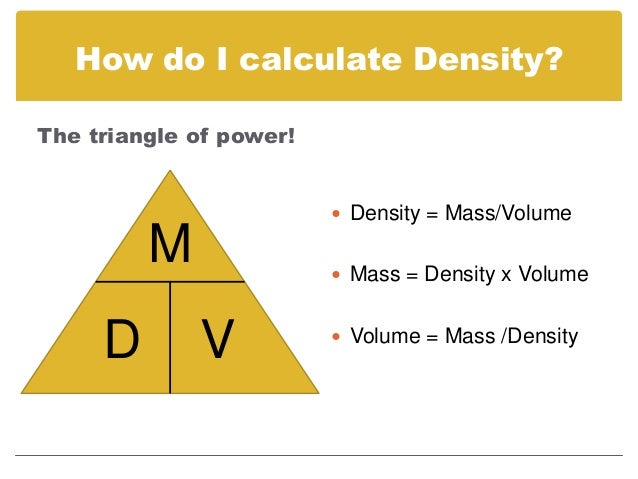
In general then, the equation to calculate density is 1/Dalloy = Mass Fraction Metal 1/Dmetal 1 + Mass Fraction Metal 2/Dmetal 2. The density of the alloy is its mass (100g) divided by its volume (11.76 cc) or 100/11.76 g/cc = 8.50 g/cc. Let's say you make a 100 g sample of this alloy, 60 g will be Sn, 40 g will be Pb. The density of tin is Dsn = 7.29 g/cc and that of lead Dpb = 11.34 g/cc. Say you want to calculate the density of an alloy of 60% by weight tin (Sn) and 40% lead (Pb). A simple explanation follows&thanks to my Indium colleague Bob Jarrett for this approach. Although I am pleased to share my Excel software that calculates solder alloy densities, being an educator, I can't help but want to share the fundamentals of how to solve this problem. The technique to calculate density of an alloy still attracts interest about a year after I first discussed it.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
